
Validation, Engineering
& Compliance Excellence.

About Valiant GMP
Who We Are
A specialist consulting firm focused on validation, engineering, and quality
compliance for the pharmaceutical and biotechnology sector.
Deep expertise across cell and gene therapies, biologics, and small‑molecule
therapeutics, supporting programs from early development through commercial
readiness.
What Sets Us Apart
Commitment to quality, transparency, and scientific rigor while providing both
strategic guidance and operational execution
GMP compliance engineered into every solution from the ground up — scaling
regulatory rigor across the full drug product lifecycle while shaping corporate
strategy, strengthening risk oversight, and enabling informed
decision-making

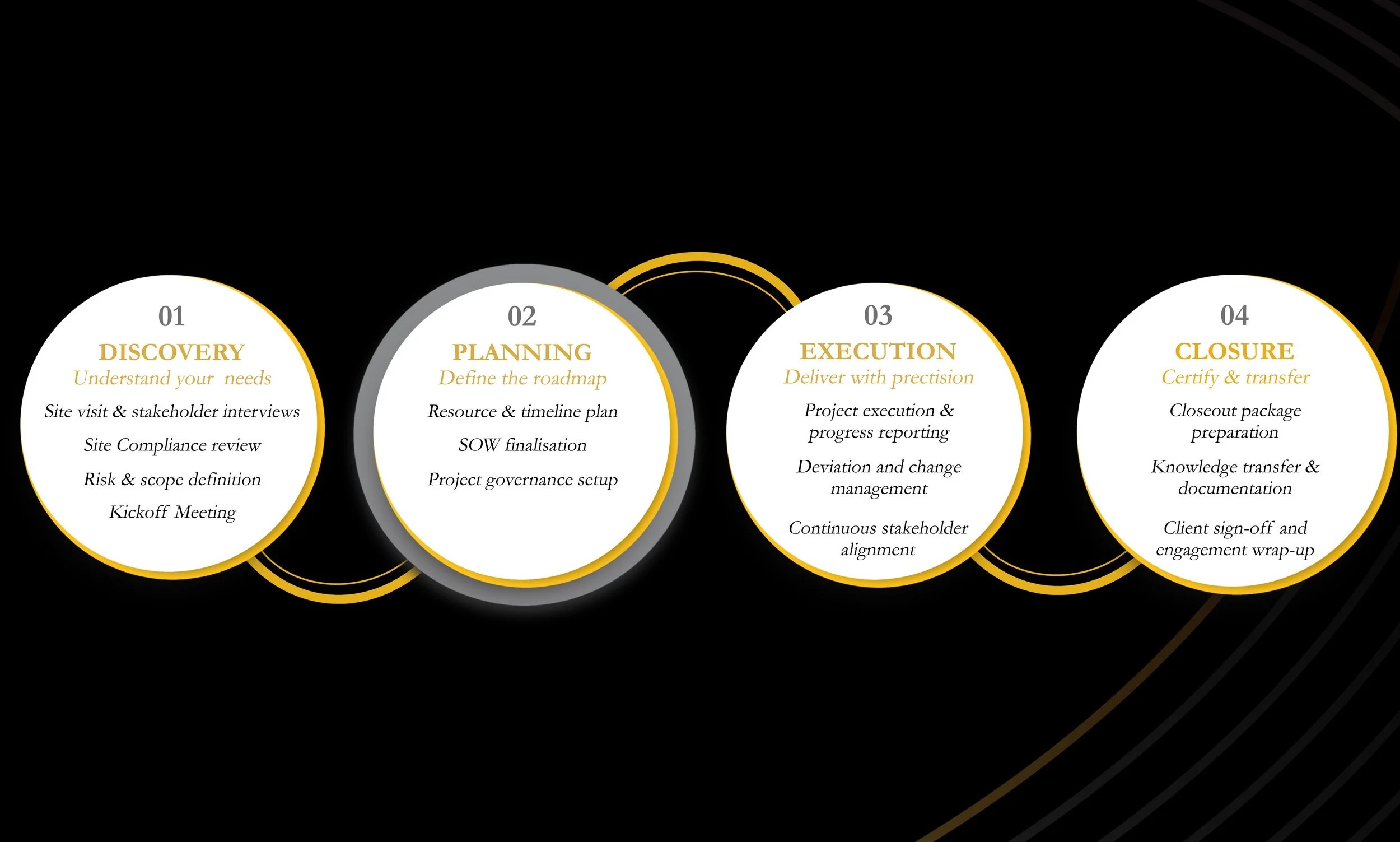
Our Engagement Approach










